  
Note that our EBAS - equation balancer and stoichiometry calculator - performs all stoichiometric calculations instantly, saving you time. of an acid or acidity of a base/Number of electron transfer per mole reaction). A mole simply represents Avogadros number (6.023 x 1023) of molecules. There are two basic methods of performing stoichiometric calculations - one is based on ratios and proportions of reagents, and the second on so-called dimensional analysis (which is not the best name, as dimensional analysis is also a much more complicated and advanced method, used for completely different purposes). Preparing Solution by Dilution:Strength Calculator. Given the equation above, we can tell the number of moles of reactants and products. While the stoichiometric information in the reaction equation is based on moles, it can be easily converted to masses - with the use of molar masses. It can also handle equations that contains fractions and decimals. It automatically balances equations and finds limiting reagents. If there is one mole of hydrogen, it will never react with more than half mole of oxygen, no matter how much oxygen is present. A comprehensive reaction stoichiometry calculator that can solve problems of all situations. It also means if there is only one mole of oxygen, it will never react with more than two moles of hydrogen - no matter how much hydrogen is present, only two moles can be consumed. Equation Balancer - Step by Step Empirical Formula - Step by Step Stoichiometry - Step by Step : Find. What is important is the fact, that the ratio of the reagents stays constant - so if we take 1 mole of the hydrogen it will react with 0.5 moles of the oxygen, if we take 4 moles of oxygen, it will react with 8 moles of hydrogen and so on - number of moles of hydrogen reacting will be always twice the number of moles of oxygen, number of moles of oxygen reacting will be always half of the number of moles of hydrogen. Stoichiometry Calculator is a free online tool that displays a balanced equation for the given chemical equation. Shows Step by Step solutions for Chemistry topics. We can read the balanced reaction equationĪs "two diatomic molecules of hydrogen reacting with one diatomic particle of oxygen, creating two particles of water" or "two moles of diatomic hydrogen reacting with one mole of diatomic oxygen, creating two moles of water". A common type of stoichiometric relationship is the mole ratio, which relates the amounts in moles of any two substances in a chemical reaction. Information presented in the balanced chemical reaction equation describes reaction, not in terms of masses of the reagents, but in terms of numbers of molecules or moles. Sometimes we want it to be able to evaluate stoichiometric amount of reagents needed for some chemical process, sometimes we want to use it for the calculation of the titration (determination) result - regardless of the reason, we have to perform a stoichiometric calculation. 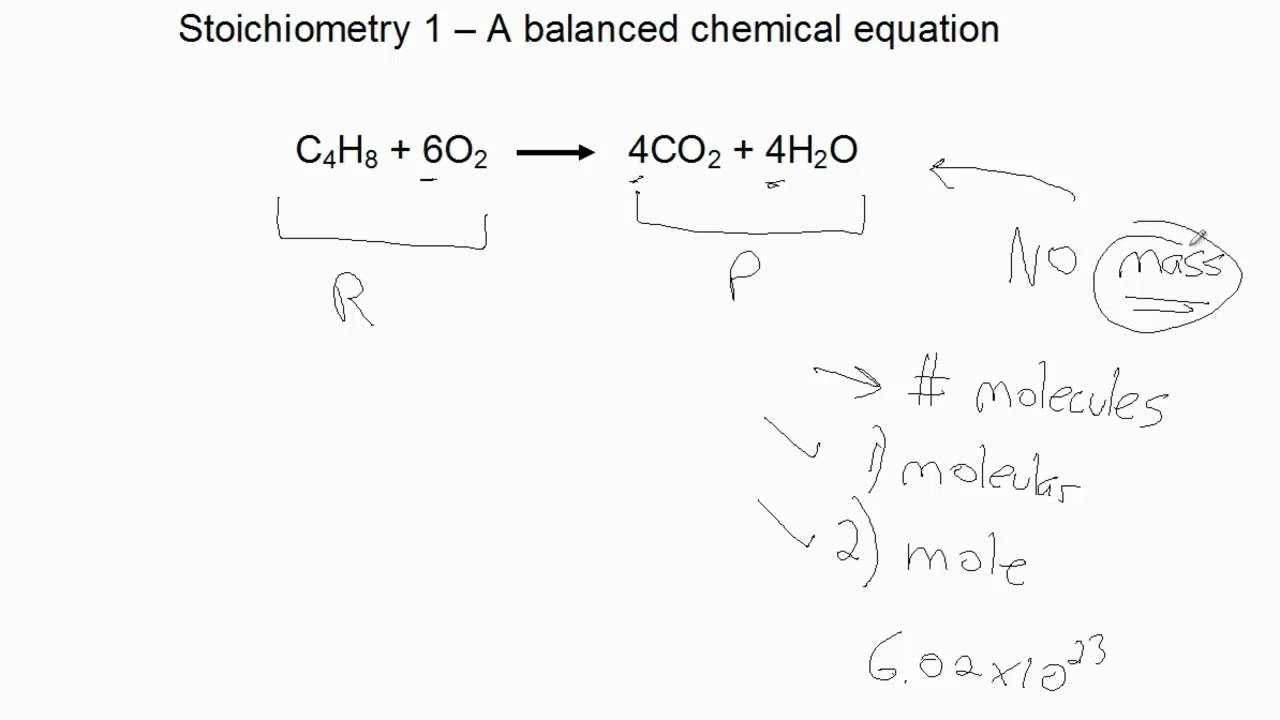
Once the reaction equation is balanced we usually want to use it to calculate amount of reagents that take place in the reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
